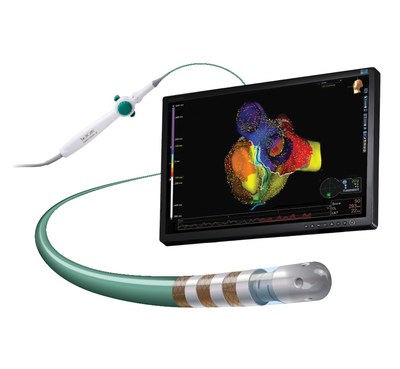
Abbott Announces CE Mark for TactiCath™ Contact Force Ablation Catheter, Sensor Enabled™
- New cardiac ablation catheter integrates with 3D mapping system to advance the treatment of atrial fibrillation
ABBOTT PARK, Ill., May 9, 2017 /PRNewswire/ -- Abbott (NYSE: ABT) today announced CE Mark of the TactiCath™ Contact Force Ablation Catheter, Sensor Enabled™, developed to make it easier for physicians to more effectively treat atrial fibrillation (AF), a condition in which the upper chambers of the heart beat too fast. When integrated with Abbott's EnSite Precision™ cardiac mapping system, physicians are able to utilize dual impedance and magnetic technologies to help more precisely model the heart. This integrated system also helps physicians determine where to apply optimal contact force (pressure) when creating a lesion during a cardiac ablation to correct a heart rhythm abnormality. The Sensor Enabled technology allows physicians to create a more detailed heart model during ablation procedures than a catheter without a sensor. The CE Mark is the latest in a line of new approvals within the company's leading atrial fibrillation portfolio.
Arrhythmias develop when electrical signals that regulate heart rhythms become disrupted or change, making the heart develop an irregular heartbeat. A patient with AF is at increased risk for stroke because the rapid heartbeat can let blood pool in the heart, which can cause clots to form and travel to the brain. To restore optimal electrical signals in the heart, ablation catheters deliver radio-frequency energy to cardiac tissue, creating small scars or lesions to areas responsible for generating arrhythmias. Not all ablation technology is the same, however, and recent research has continued to show the importance of applying optimal contact force to the heart during ablation procedures. Too much pressure applied to heart tissue can result in complications, yet if not enough pressure is applied the lesion may not be effective enough to stop the erratic signals in the heart.
"The TactiCath™ Contact Force Ablation Catheter, Sensor Enabled™ along with the EnSite Precision™ cardiac mapping system create a powerful combination for more precisely treating patients with cardiac arrhythmias," said Martin Lowe, M.D., clinical director of cardiac electrophysiology at Bart's Heart Centre at St. Bartholomew's Hospital in London. "Feeling confident in the accuracy of the contact force reading allows me to target the optimal pressure for creating effective and safe lesions."
The TactiCath™ Contact Force Ablation Catheter, Sensor Enabled™, is a significant update to the TactiCath™ Contact Force Catheter family – which is the most-studied Contact Force ablation catheter platform to date and has unmatched clinical evidence based on number of completed prospective, protocol driven, industry-sponsored, registered studies on Contact Force technology. The clinical evidence show use of optimal Contact Force guided AF ablation with the TactiCath™ Quartz catheter resulted in clinical success in 85.5% of patients and fewer post-ablation clinical events, translating to a 15% reduction in post-ablation management costs ($3,402 savings per patient) in the year after ablation vs. patients treated with a non-contact force ablation catheter.
The magnetic sensor of the TactiCath™ Contact Force Ablation Catheter, Sensor Enabled™ integrates with the Abbott EnSite Precision™ cardiac mapping system, providing the ability to collect both magnetic and impedance (resistance) data so physicians can create more life-like models of the heart. It also uses the Abbott FlexAbility™ catheter platform's advanced ergonomic handle-shaft combination, which is designed for reach, maneuverability and 1:1 torque.
When utilized with the EnSite Precision™ cardiac mapping system, the TactiCath™ Contact Force Ablation Catheter, Sensor Enabled™ can improve workflow efficiency in the lab. The system enables automated guidance of lesion marking, verification of the ablation catheter stability, the ability to automatically record the precise location of the catheter tip during radiofrequency energy application, the capability to add mapping points based on Contact Force and the ability to review and identify any potential gaps by viewing specific lesions from the display list.
"The goal in developing the TactiCath™ Contact Force Ablation Catheter, Sensor Enabled™ was to provide the most innovative solution for treating atrial fibrillation and lead the way in clinical outcomes for patients with cardiac arrhythmias – even during long and complex ablation procedures," said Srijoy Mahapatra, M.D., FHRS, medical director of Abbott's electrophysiology business. "Integration with the EnSite Precision™ cardiac mapping system provides an unprecedented opportunity to help patients suffering with atrial fibrillation."
Currently the TactiCath™ Contact Force Ablation Catheter, Sensor Enabled™ is available in select markets in Europe, with full market release expected in the third quarter. Abbott is pursuing approval in the U.S.
About Abbott:
At Abbott, we're committed to helping people live their best possible life through the power of health. For more than 125 years, we've brought new products and technologies to the world -- in nutrition, diagnostics, medical devices and branded generic pharmaceuticals -- that create more possibilities for more people at all stages of life. Today, 94,000 of us are working to help people live not just longer, but better, in the more than 150 countries we serve.
Connect with us at www.abbott.com, on Facebook at www.facebook.com/Abbott and on Twitter @AbbottNews and @AbbottGlobal.
SOURCE Abbott
For further information: Abbott Media: Gina O'Connell, 651-756-3380 goconnell02@sjm.com, Abbott Financial: J.C. Weigelt, 651-756-4347, jweigelt@sjm.com
 BACK TO PRESS RELEASES
BACK TO PRESS RELEASES